Patient-reported outcomes: A new era in clinical research Deshpande PR, Rajan S, Sudeepthi B L, Abdul Nazir C P - Perspect Clin Res

Patient-reported outcomes: A new era in clinical research – topic of research paper in Clinical medicine. Download scholarly article PDF and read for free on CyberLeninka open science hub.

Cardiovascular outcomes in adults with hypertension with evening versus morning dosing of usual antihypertensives in the UK (TIME study): a prospective, randomised, open-label, blinded-endpoint clinical trial - The Lancet
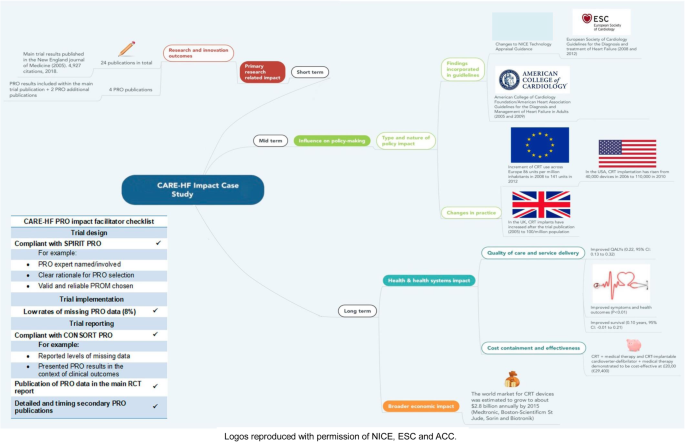
The impact of patient-reported outcome (PRO) data from clinical trials: a systematic review and critical analysis | Health and Quality of Life Outcomes | Full Text

Patient-reported outcomes: A new era in clinical research Deshpande PR, Rajan S, Sudeepthi B L, Abdul Nazir C P - Perspect Clin Res
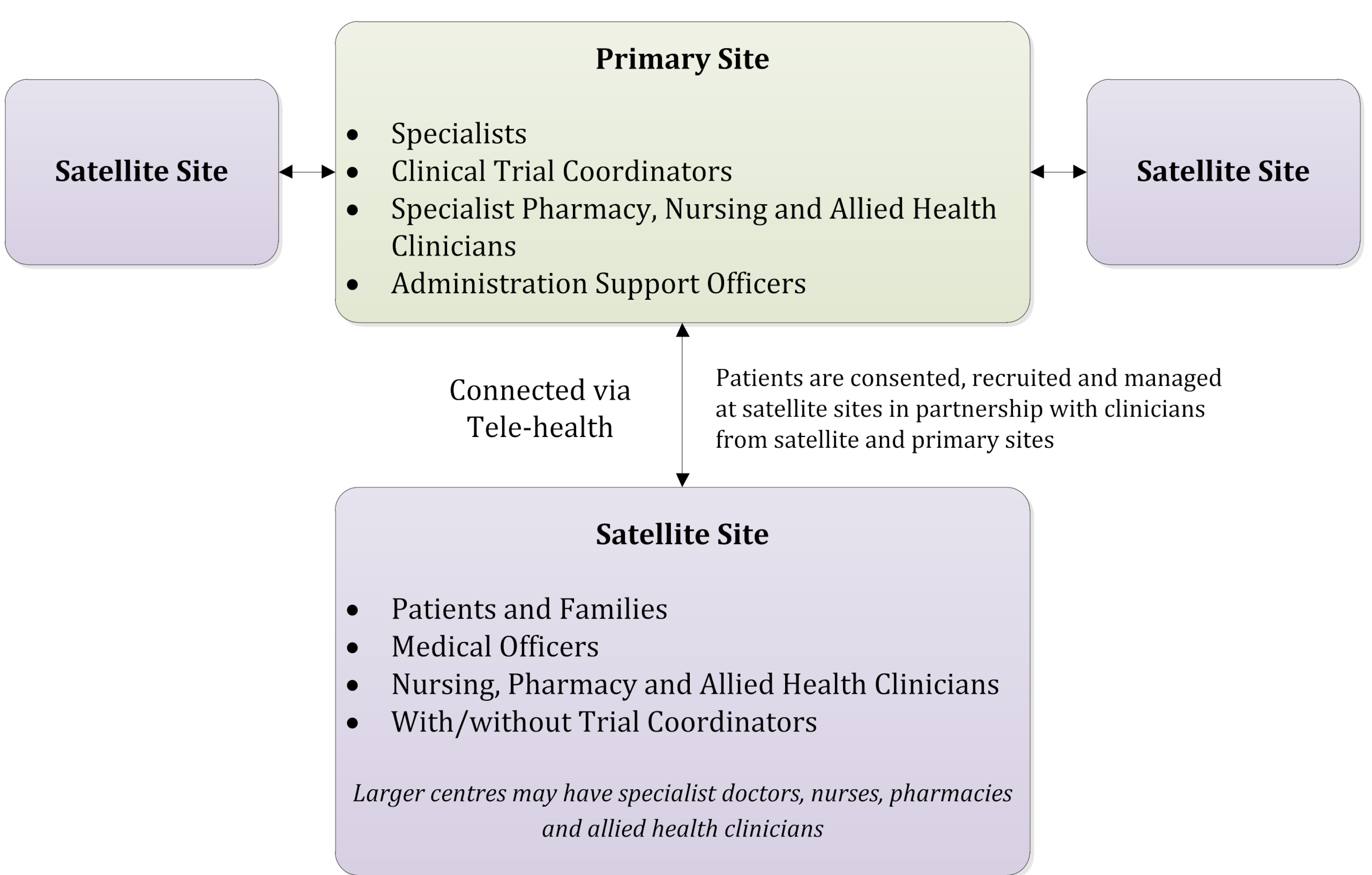
Current Oncology | Free Full-Text | CRAFT—A Proposed Framework for Decentralized Clinical Trials Participation in Canada
Patient-reported outcomes for diabetes and hypertension care in low- and middle-income countries: A scoping review | PLOS ONE
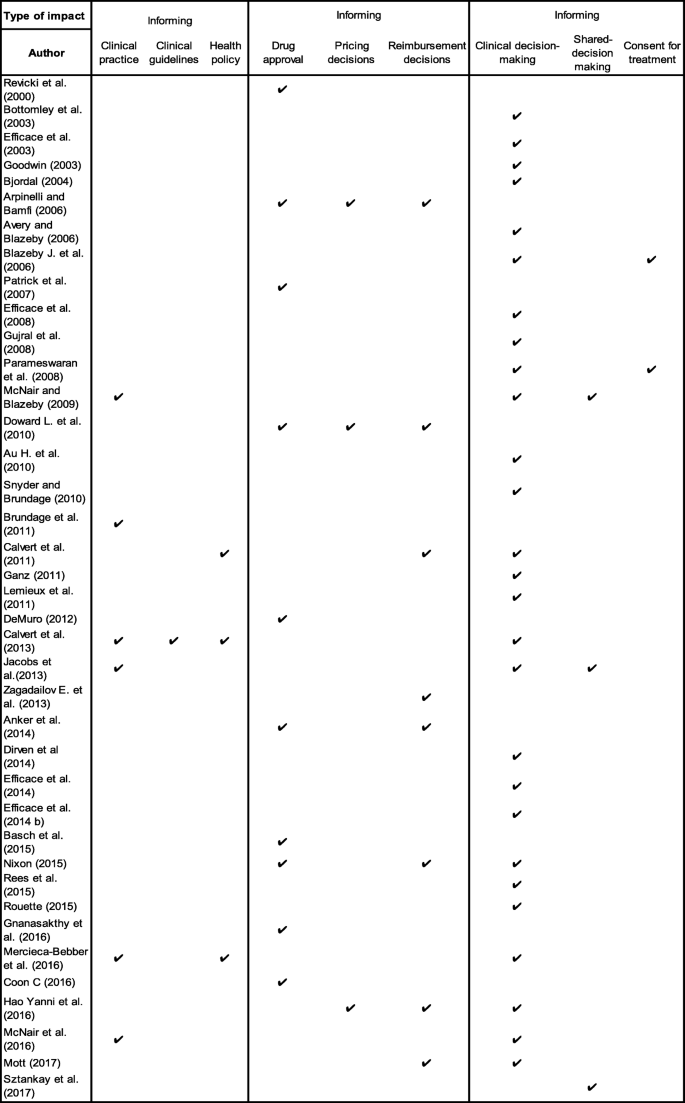
The impact of patient-reported outcome (PRO) data from clinical trials: a systematic review and critical analysis | Health and Quality of Life Outcomes | Full Text
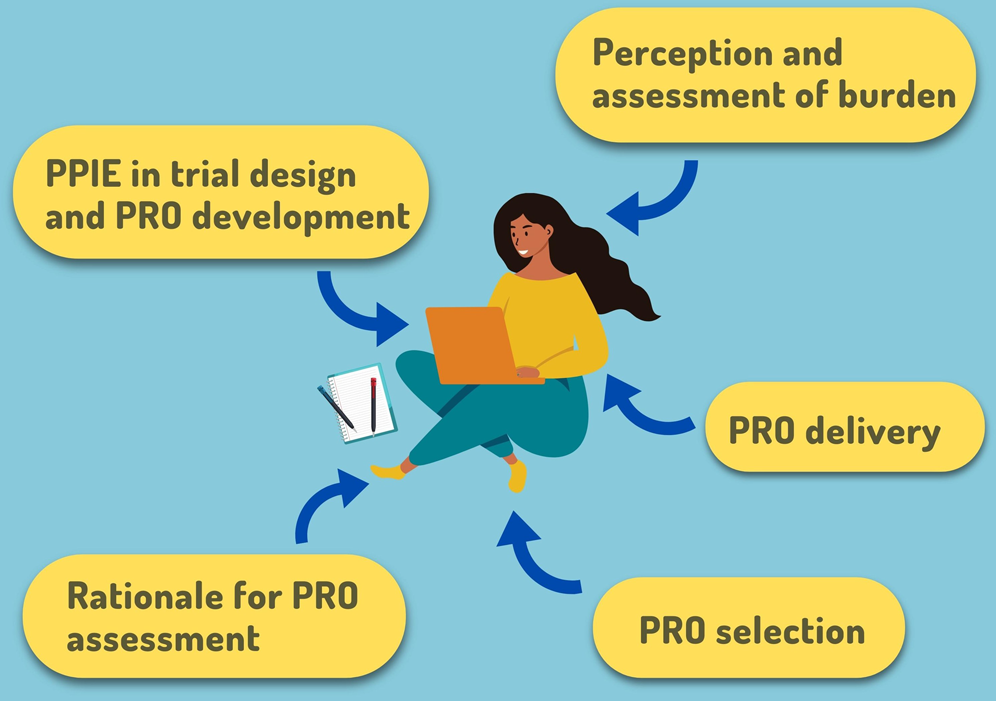
Key considerations to reduce or address respondent burden in patient-reported outcome (PRO) data collection | Nature Communications

Patient-reported outcomes with first-line nivolumab plus cabozantinib versus sunitinib in patients with advanced renal cell carcinoma treated in CheckMate 9ER: an open-label, randomised, phase 3 trial - The Lancet Oncology
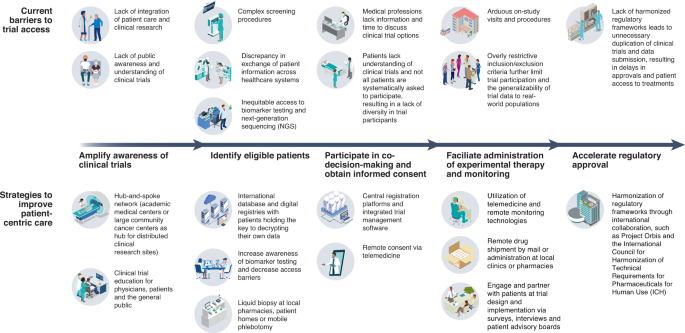
Reimagining patient-centric cancer clinical trials: a multi-stakeholder international coalition | Nature Medicine